 
A liquid is more ordered than a gas, therefore entropy has decreased. This is as we would expect since we have gone from two gases to a gas and a liquid. For example, the production of ammonia: In this case, there is a decrease in entropy during the forward reaction because there are fewer gas molecules than you had to start with. Reactions involving a decrease in the number of gas molecules. Notice the negative sign, this indicates that entropy has decreased in the reaction. Entropy will increase during such a reaction, because of the increased disorder. Entropy Chemical and physical changes in a system may be accompanied by either an increase or a decrease in the disorder of the system, corresponding to an increase in entropy (S > 0) or a decrease in entropy (S < 0), respectively. Calculate the entropy change for the reaction using the equation. 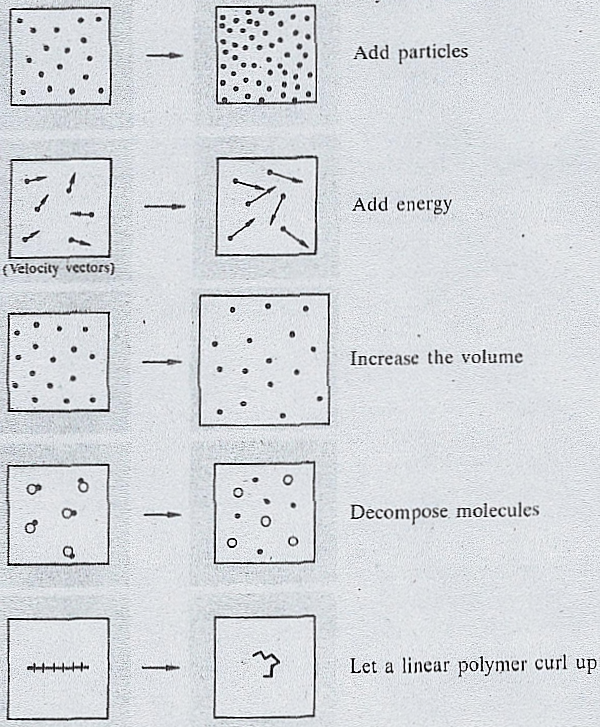
You can work out the entropy of the reactants by adding up the entropy values for each reactant in a reactionģ. You can work out the entropy of the products by adding up the entropy values for each product in a reactionĢ. This is just the difference in entropy between the sum of the products and the sum of the reactants.Įxample: Calculate the entropy change for this reaction:ġ. The formula for calculating ΔS is as follows: They are typically found at standard conditions. In a chemical reactions, products and reactants have different entropies – there is an entropy change.įor reactions with a negative entropy change, reactants will have a higher entropy than products.įor reactions with a positive entropy change, products will have a higher entropy than reactants.Įntropy values have been determined and can be found in databases. Therefore, substance will naturally move in order to increase their entropy. The more disordered a system is, the higher its entropy and the more stable it is. Notice that there isnt very big jump in entropy when ice. From them we can draw some general conclusions. The entropy increases as the molecules become more disordered as you go from solid to liquid to gas. When in a reaction the number of moles of product formed is greater than the number of moles of reactants, there is an increase in entropy. Below we have various examples of entropy change during various processes. Number of particles – For an increased number of particles there is and increased number of ways in which these can be arranged and therefore an increased entropy. Dissolving – When a solid dissolves in a solvent, its entropy increases because of the free movement of the dissolved particles.ģ. Thermodynamic – Introduction to EntropyĢ. This is because the arrangement of particles is more random in gases than solids. The tendency of increasing entropy is to fill space, to spread out particles and energy in all possible ways. An increase in temperature means that the particles of the substance have greater kinetic energy. State of Matter – Generally, solids have lower entropy than liquids, which have lower entropy than gases. Entropy increases as temperature increases. The more disordered a system is, the higher (the more positive) the value of entropy.ġ. 
Thermodynamic - Introduction to Entropy (A-Level Chemistry) Introduction to Entropy Entropy Key TermsĮntropy can be defined as the randomness or dispersal of energy of a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
